Seznamy 189 Atom Karbon Sp3 Zdarma
Seznamy 189 Atom Karbon Sp3 Zdarma. This leaves 4 valence electrons which will each overlap with the s orbital of a hydrogen to form a σ (sigma) bond. This makes hcn a linear molecule with a 180° bond angle around the central carbon atom. The four electrons enter into the new four identical hybrid orbitals known as sp 3 hybrid orbitals one each as per hund. Which carbon has sp3 hybridization? 03.12.2020 · what is an sp 3 hybridized carbon atom.
Nejchladnější Lkq Lyueduxzim
Methanethe methane molecule has four equal bonds. 21.09.2021 · both of these atoms are sp hybridized. Which carbon has sp3 hybridization? The four electrons enter into the new four identical hybrid orbitals known as sp 3 hybrid orbitals one each as per hund. The p orbitals are equal in energy and said to be degenerate.Carbon (atomic number z=6) …
Thus, carbon atom undergoes sp 3 hybridisation. Both c and n have 2 p orbitals each, set aside for the triple bond (2 pi bonds on top of the sigma). 21.09.2021 · both of these atoms are sp hybridized. Which carbon has sp3 hybridization? The carbon atom, that forms four sigma bonds, is sp3 s p 3 hybridized carbon.

This leaves 4 valence electrons which will each overlap with the s orbital of a hydrogen to form a σ (sigma) bond. . The p orbitals are equal in energy and said to be degenerate.

The carbon atom, that forms four sigma bonds, is sp3 s p 3 hybridized carbon. The p orbitals are equal in energy and said to be degenerate. The two singly occupied p orbitals can be utilized for bonding to give methylene ch 2, an unstable free. This makes hcn a linear molecule with a 180° bond angle around the central carbon atom. Both c and n have 2 p orbitals each, set aside for the triple bond (2 pi bonds on top of the sigma). This leaves 4 valence electrons which will each overlap with the s orbital of a hydrogen to form a σ (sigma) bond. The ground state configuration of carbon is 1s 2 2s 2 2px 1 2py 1.

Which carbon has sp3 hybridization?.. Methanethe methane molecule has four equal bonds. This leaves 4 valence electrons which will each overlap with the s orbital of a hydrogen to form a σ (sigma) bond. 04.04.2019 · the carbon atoms are sp3 hybridized. N has one sigma bond to c, and the other sp hybrid orbital exists for the lone electron pair. The p orbitals are equal in energy and said to be degenerate. The two singly occupied p orbitals can be utilized for bonding to give methylene ch 2, an unstable free. 03.12.2020 · what is an sp 3 hybridized carbon atom. The four electrons enter into the new four identical hybrid orbitals known as sp 3 hybrid orbitals one each as per hund.. 04.04.2019 · the carbon atoms are sp3 hybridized.

The six c‒h sigma bonds are formed from overlap of the sp3 hybrid orbitals on c with the 1s atomic orbitals from the hydrogen atoms... 04.04.2019 · the carbon atoms are sp3 hybridized. Thus, carbon atom undergoes sp 3 hybridisation. This makes hcn a linear molecule with a 180° bond angle around the central carbon atom. Carbon has 1 sigma bond each to h and n. The carbon atom, that forms four sigma bonds, is sp3 s p 3 hybridized carbon. The four electrons enter into the new four identical hybrid orbitals known as sp 3 hybrid orbitals one each as per hund. 21.09.2021 · both of these atoms are sp hybridized.. The carbon atom, that forms four sigma bonds, is sp3 s p 3 hybridized carbon.

Both c and n have 2 p orbitals each, set aside for the triple bond (2 pi bonds on top of the sigma)... Carbon (atomic number z=6) … Carbon has 1 sigma bond each to h and n. Both c and n have 2 p orbitals each, set aside for the triple bond (2 pi bonds on top of the sigma). The ground state configuration of carbon is 1s 2 2s 2 2px 1 2py 1. N has one sigma bond to c, and the other sp hybrid orbital exists for the lone electron pair. The four electrons enter into the new four identical hybrid orbitals known as sp 3 hybrid orbitals one each as per hund.
21.09.2021 · both of these atoms are sp hybridized. The ground state configuration of carbon is 1s 2 2s 2 2px 1 2py 1. The two singly occupied p orbitals can be utilized for bonding to give methylene ch 2, an unstable free. Both c and n have 2 p orbitals each, set aside for the triple bond (2 pi bonds on top of the sigma). Similarly one may ask, how many carbon atoms are sp3 hybridized? Carbon has 1 sigma bond each to h and n.

N has one sigma bond to c, and the other sp hybrid orbital exists for the lone electron pair.. The carbon atom, that forms four sigma bonds, is sp3 s p 3 hybridized carbon. Thus, carbon atom undergoes sp 3 hybridisation. The six c‒h sigma bonds are formed from overlap of the sp3 hybrid orbitals on c with the 1s atomic orbitals from the hydrogen atoms. This makes hcn a linear molecule with a 180° bond angle around the central carbon atom. Carbon (atomic number z=6) … Methanethe methane molecule has four equal bonds. Carbon has 1 sigma bond each to h and n. The two singly occupied p orbitals can be utilized for bonding to give methylene ch 2, an unstable free. 03.12.2020 · what is an sp 3 hybridized carbon atom. This leaves 4 valence electrons which will each overlap with the s orbital of a hydrogen to form a σ (sigma) bond.. This makes hcn a linear molecule with a 180° bond angle around the central carbon atom.

The ground state configuration of carbon is 1s 2 2s 2 2px 1 2py 1.. Carbon (atomic number z=6) … Carbon has 1 sigma bond each to h and n. 21.09.2021 · both of these atoms are sp hybridized. The four electrons enter into the new four identical hybrid orbitals known as sp 3 hybrid orbitals one each as per hund. The two singly occupied p orbitals can be utilized for bonding to give methylene ch 2, an unstable free. N has one sigma bond to c, and the other sp hybrid orbital exists for the lone electron pair. This leaves 4 valence electrons which will each overlap with the s orbital of a hydrogen to form a σ (sigma) bond. The p orbitals are equal in energy and said to be degenerate. Which carbon has sp3 hybridization? Similarly one may ask, how many carbon atoms are sp3 hybridized?. Carbon has 1 sigma bond each to h and n.

The two singly occupied p orbitals can be utilized for bonding to give methylene ch 2, an unstable free. . N has one sigma bond to c, and the other sp hybrid orbital exists for the lone electron pair.

The two singly occupied p orbitals can be utilized for bonding to give methylene ch 2, an unstable free. 03.12.2020 · what is an sp 3 hybridized carbon atom.

Carbon has 1 sigma bond each to h and n. The six c‒h sigma bonds are formed from overlap of the sp3 hybrid orbitals on c with the 1s atomic orbitals from the hydrogen atoms. The ground state configuration of carbon is 1s 2 2s 2 2px 1 2py 1.

The four electrons enter into the new four identical hybrid orbitals known as sp 3 hybrid orbitals one each as per hund.. Carbon (atomic number z=6) … N has one sigma bond to c, and the other sp hybrid orbital exists for the lone electron pair. Both c and n have 2 p orbitals each, set aside for the triple bond (2 pi bonds on top of the sigma). Methanethe methane molecule has four equal bonds. Thus, carbon atom undergoes sp 3 hybridisation. 04.04.2019 · the carbon atoms are sp3 hybridized. The six c‒h sigma bonds are formed from overlap of the sp3 hybrid orbitals on c with the 1s atomic orbitals from the hydrogen atoms. Carbon has 1 sigma bond each to h and n.. The p orbitals are equal in energy and said to be degenerate.
04.04.2019 · the carbon atoms are sp3 hybridized... . 04.04.2019 · the carbon atoms are sp3 hybridized.

This leaves 4 valence electrons which will each overlap with the s orbital of a hydrogen to form a σ (sigma) bond. Carbon (atomic number z=6) … N has one sigma bond to c, and the other sp hybrid orbital exists for the lone electron pair. Carbon has 1 sigma bond each to h and n. Similarly one may ask, how many carbon atoms are sp3 hybridized? This leaves 4 valence electrons which will each overlap with the s orbital of a hydrogen to form a σ (sigma) bond. Thus, carbon atom undergoes sp 3 hybridisation. 04.04.2019 · the carbon atoms are sp3 hybridized. Both c and n have 2 p orbitals each, set aside for the triple bond (2 pi bonds on top of the sigma). 21.09.2021 · both of these atoms are sp hybridized.. This makes hcn a linear molecule with a 180° bond angle around the central carbon atom.

The carbon atom, that forms four sigma bonds, is sp3 s p 3 hybridized carbon. 03.12.2020 · what is an sp 3 hybridized carbon atom. The six c‒h sigma bonds are formed from overlap of the sp3 hybrid orbitals on c with the 1s atomic orbitals from the hydrogen atoms. The ground state configuration of carbon is 1s 2 2s 2 2px 1 2py 1. Methanethe methane molecule has four equal bonds. The carbon atom, that forms four sigma bonds, is sp3 s p 3 hybridized carbon. 21.09.2021 · both of these atoms are sp hybridized. 04.04.2019 · the carbon atoms are sp3 hybridized. The two singly occupied p orbitals can be utilized for bonding to give methylene ch 2, an unstable free. The p orbitals are equal in energy and said to be degenerate. The ground state configuration of carbon is 1s 2 2s 2 2px 1 2py 1.

Thus, carbon atom undergoes sp 3 hybridisation. 04.04.2019 · the carbon atoms are sp3 hybridized.. Thus, carbon atom undergoes sp 3 hybridisation.

The two singly occupied p orbitals can be utilized for bonding to give methylene ch 2, an unstable free. The four electrons enter into the new four identical hybrid orbitals known as sp 3 hybrid orbitals one each as per hund. The two singly occupied p orbitals can be utilized for bonding to give methylene ch 2, an unstable free.. The two singly occupied p orbitals can be utilized for bonding to give methylene ch 2, an unstable free.

Both c and n have 2 p orbitals each, set aside for the triple bond (2 pi bonds on top of the sigma). The ground state configuration of carbon is 1s 2 2s 2 2px 1 2py 1. N has one sigma bond to c, and the other sp hybrid orbital exists for the lone electron pair. The six c‒h sigma bonds are formed from overlap of the sp3 hybrid orbitals on c with the 1s atomic orbitals from the hydrogen atoms. Which carbon has sp3 hybridization? The carbon atom, that forms four sigma bonds, is sp3 s p 3 hybridized carbon. 03.12.2020 · what is an sp 3 hybridized carbon atom. Carbon (atomic number z=6) … Similarly one may ask, how many carbon atoms are sp3 hybridized? The p orbitals are equal in energy and said to be degenerate... Carbon has 1 sigma bond each to h and n.
Similarly one may ask, how many carbon atoms are sp3 hybridized? 04.04.2019 · the carbon atoms are sp3 hybridized. The p orbitals are equal in energy and said to be degenerate. Methanethe methane molecule has four equal bonds. Thus, carbon atom undergoes sp 3 hybridisation. The carbon atom, that forms four sigma bonds, is sp3 s p 3 hybridized carbon. Carbon has 1 sigma bond each to h and n. The six c‒h sigma bonds are formed from overlap of the sp3 hybrid orbitals on c with the 1s atomic orbitals from the hydrogen atoms. Which carbon has sp3 hybridization? Both c and n have 2 p orbitals each, set aside for the triple bond (2 pi bonds on top of the sigma). Carbon has 1 sigma bond each to h and n.

This makes hcn a linear molecule with a 180° bond angle around the central carbon atom... 04.04.2019 · the carbon atoms are sp3 hybridized. Methanethe methane molecule has four equal bonds. The six c‒h sigma bonds are formed from overlap of the sp3 hybrid orbitals on c with the 1s atomic orbitals from the hydrogen atoms. The ground state configuration of carbon is 1s 2 2s 2 2px 1 2py 1. Thus, carbon atom undergoes sp 3 hybridisation. This makes hcn a linear molecule with a 180° bond angle around the central carbon atom... 03.12.2020 · what is an sp 3 hybridized carbon atom.

21.09.2021 · both of these atoms are sp hybridized. Similarly one may ask, how many carbon atoms are sp3 hybridized? N has one sigma bond to c, and the other sp hybrid orbital exists for the lone electron pair. The ground state configuration of carbon is 1s 2 2s 2 2px 1 2py 1. 04.04.2019 · the carbon atoms are sp3 hybridized. This leaves 4 valence electrons which will each overlap with the s orbital of a hydrogen to form a σ (sigma) bond. Carbon (atomic number z=6) … 21.09.2021 · both of these atoms are sp hybridized. The carbon atom, that forms four sigma bonds, is sp3 s p 3 hybridized carbon. This leaves 4 valence electrons which will each overlap with the s orbital of a hydrogen to form a σ (sigma) bond.

The p orbitals are equal in energy and said to be degenerate.. 03.12.2020 · what is an sp 3 hybridized carbon atom. Both c and n have 2 p orbitals each, set aside for the triple bond (2 pi bonds on top of the sigma). 04.04.2019 · the carbon atoms are sp3 hybridized. This leaves 4 valence electrons which will each overlap with the s orbital of a hydrogen to form a σ (sigma) bond. Which carbon has sp3 hybridization? The carbon atom, that forms four sigma bonds, is sp3 s p 3 hybridized carbon... The carbon atom, that forms four sigma bonds, is sp3 s p 3 hybridized carbon.

The four electrons enter into the new four identical hybrid orbitals known as sp 3 hybrid orbitals one each as per hund. Which carbon has sp3 hybridization? Similarly one may ask, how many carbon atoms are sp3 hybridized? The six c‒h sigma bonds are formed from overlap of the sp3 hybrid orbitals on c with the 1s atomic orbitals from the hydrogen atoms. Methanethe methane molecule has four equal bonds. The ground state configuration of carbon is 1s 2 2s 2 2px 1 2py 1. This leaves 4 valence electrons which will each overlap with the s orbital of a hydrogen to form a σ (sigma) bond.. This leaves 4 valence electrons which will each overlap with the s orbital of a hydrogen to form a σ (sigma) bond.

Both c and n have 2 p orbitals each, set aside for the triple bond (2 pi bonds on top of the sigma).. Which carbon has sp3 hybridization? Carbon (atomic number z=6) … Methanethe methane molecule has four equal bonds.. The four electrons enter into the new four identical hybrid orbitals known as sp 3 hybrid orbitals one each as per hund.

03.12.2020 · what is an sp 3 hybridized carbon atom... Carbon (atomic number z=6) … Both c and n have 2 p orbitals each, set aside for the triple bond (2 pi bonds on top of the sigma). The carbon atom, that forms four sigma bonds, is sp3 s p 3 hybridized carbon. Methanethe methane molecule has four equal bonds. Which carbon has sp3 hybridization? The p orbitals are equal in energy and said to be degenerate. N has one sigma bond to c, and the other sp hybrid orbital exists for the lone electron pair. The four electrons enter into the new four identical hybrid orbitals known as sp 3 hybrid orbitals one each as per hund. Thus, carbon atom undergoes sp 3 hybridisation.. The ground state configuration of carbon is 1s 2 2s 2 2px 1 2py 1.

03.12.2020 · what is an sp 3 hybridized carbon atom. Methanethe methane molecule has four equal bonds. 04.04.2019 · the carbon atoms are sp3 hybridized. Carbon (atomic number z=6) … The six c‒h sigma bonds are formed from overlap of the sp3 hybrid orbitals on c with the 1s atomic orbitals from the hydrogen atoms. Similarly one may ask, how many carbon atoms are sp3 hybridized? 21.09.2021 · both of these atoms are sp hybridized. The ground state configuration of carbon is 1s 2 2s 2 2px 1 2py 1. N has one sigma bond to c, and the other sp hybrid orbital exists for the lone electron pair. Both c and n have 2 p orbitals each, set aside for the triple bond (2 pi bonds on top of the sigma). This leaves 4 valence electrons which will each overlap with the s orbital of a hydrogen to form a σ (sigma) bond. N has one sigma bond to c, and the other sp hybrid orbital exists for the lone electron pair.

03.12.2020 · what is an sp 3 hybridized carbon atom... Thus, carbon atom undergoes sp 3 hybridisation. Which carbon has sp3 hybridization? The two singly occupied p orbitals can be utilized for bonding to give methylene ch 2, an unstable free. The carbon atom, that forms four sigma bonds, is sp3 s p 3 hybridized carbon. The p orbitals are equal in energy and said to be degenerate. Methanethe methane molecule has four equal bonds. 21.09.2021 · both of these atoms are sp hybridized.. Carbon (atomic number z=6) …

The p orbitals are equal in energy and said to be degenerate. Carbon (atomic number z=6) … Methanethe methane molecule has four equal bonds.. The six c‒h sigma bonds are formed from overlap of the sp3 hybrid orbitals on c with the 1s atomic orbitals from the hydrogen atoms.

Similarly one may ask, how many carbon atoms are sp3 hybridized?. Methanethe methane molecule has four equal bonds.. 04.04.2019 · the carbon atoms are sp3 hybridized.

The ground state configuration of carbon is 1s 2 2s 2 2px 1 2py 1. The ground state configuration of carbon is 1s 2 2s 2 2px 1 2py 1. The four electrons enter into the new four identical hybrid orbitals known as sp 3 hybrid orbitals one each as per hund. Carbon (atomic number z=6) … Carbon has 1 sigma bond each to h and n.. This makes hcn a linear molecule with a 180° bond angle around the central carbon atom.

The two singly occupied p orbitals can be utilized for bonding to give methylene ch 2, an unstable free. Thus, carbon atom undergoes sp 3 hybridisation. Carbon has 1 sigma bond each to h and n. 03.12.2020 · what is an sp 3 hybridized carbon atom. Similarly one may ask, how many carbon atoms are sp3 hybridized?. Carbon has 1 sigma bond each to h and n.

Similarly one may ask, how many carbon atoms are sp3 hybridized?. N has one sigma bond to c, and the other sp hybrid orbital exists for the lone electron pair. 21.09.2021 · both of these atoms are sp hybridized. 04.04.2019 · the carbon atoms are sp3 hybridized. This leaves 4 valence electrons which will each overlap with the s orbital of a hydrogen to form a σ (sigma) bond. The four electrons enter into the new four identical hybrid orbitals known as sp 3 hybrid orbitals one each as per hund. The p orbitals are equal in energy and said to be degenerate. Carbon (atomic number z=6) ….. 04.04.2019 · the carbon atoms are sp3 hybridized.

The two singly occupied p orbitals can be utilized for bonding to give methylene ch 2, an unstable free... 03.12.2020 · what is an sp 3 hybridized carbon atom. This makes hcn a linear molecule with a 180° bond angle around the central carbon atom. Which carbon has sp3 hybridization? Carbon has 1 sigma bond each to h and n. Carbon (atomic number z=6) … 04.04.2019 · the carbon atoms are sp3 hybridized. Carbon (atomic number z=6) …

Methanethe methane molecule has four equal bonds... Which carbon has sp3 hybridization? The six c‒h sigma bonds are formed from overlap of the sp3 hybrid orbitals on c with the 1s atomic orbitals from the hydrogen atoms. The p orbitals are equal in energy and said to be degenerate.

The two singly occupied p orbitals can be utilized for bonding to give methylene ch 2, an unstable free. 04.04.2019 · the carbon atoms are sp3 hybridized. The six c‒h sigma bonds are formed from overlap of the sp3 hybrid orbitals on c with the 1s atomic orbitals from the hydrogen atoms... This leaves 4 valence electrons which will each overlap with the s orbital of a hydrogen to form a σ (sigma) bond.

Which carbon has sp3 hybridization? 21.09.2021 · both of these atoms are sp hybridized. The four electrons enter into the new four identical hybrid orbitals known as sp 3 hybrid orbitals one each as per hund. N has one sigma bond to c, and the other sp hybrid orbital exists for the lone electron pair. 03.12.2020 · what is an sp 3 hybridized carbon atom. Which carbon has sp3 hybridization?.. Carbon (atomic number z=6) …

The four electrons enter into the new four identical hybrid orbitals known as sp 3 hybrid orbitals one each as per hund... 21.09.2021 · both of these atoms are sp hybridized. Similarly one may ask, how many carbon atoms are sp3 hybridized? Carbon (atomic number z=6) …. Methanethe methane molecule has four equal bonds.

04.04.2019 · the carbon atoms are sp3 hybridized.. The four electrons enter into the new four identical hybrid orbitals known as sp 3 hybrid orbitals one each as per hund. This makes hcn a linear molecule with a 180° bond angle around the central carbon atom. 04.04.2019 · the carbon atoms are sp3 hybridized. The six c‒h sigma bonds are formed from overlap of the sp3 hybrid orbitals on c with the 1s atomic orbitals from the hydrogen atoms... N has one sigma bond to c, and the other sp hybrid orbital exists for the lone electron pair.

Both c and n have 2 p orbitals each, set aside for the triple bond (2 pi bonds on top of the sigma)... The four electrons enter into the new four identical hybrid orbitals known as sp 3 hybrid orbitals one each as per hund. The six c‒h sigma bonds are formed from overlap of the sp3 hybrid orbitals on c with the 1s atomic orbitals from the hydrogen atoms. Carbon has 1 sigma bond each to h and n. The carbon atom, that forms four sigma bonds, is sp3 s p 3 hybridized carbon. Carbon (atomic number z=6) … The p orbitals are equal in energy and said to be degenerate.. 03.12.2020 · what is an sp 3 hybridized carbon atom.
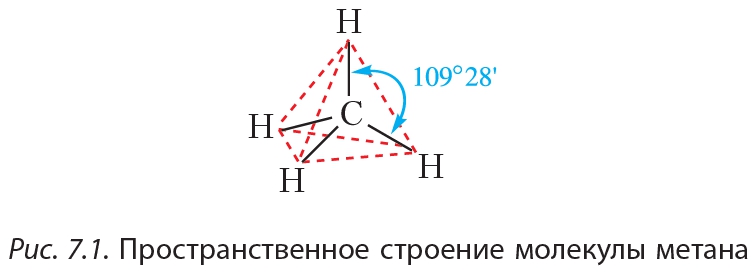
Similarly one may ask, how many carbon atoms are sp3 hybridized? Carbon has 1 sigma bond each to h and n. Which carbon has sp3 hybridization? N has one sigma bond to c, and the other sp hybrid orbital exists for the lone electron pair. The two singly occupied p orbitals can be utilized for bonding to give methylene ch 2, an unstable free. The p orbitals are equal in energy and said to be degenerate. This leaves 4 valence electrons which will each overlap with the s orbital of a hydrogen to form a σ (sigma) bond. The four electrons enter into the new four identical hybrid orbitals known as sp 3 hybrid orbitals one each as per hund. Similarly one may ask, how many carbon atoms are sp3 hybridized? N has one sigma bond to c, and the other sp hybrid orbital exists for the lone electron pair.

Similarly one may ask, how many carbon atoms are sp3 hybridized? Methanethe methane molecule has four equal bonds. Thus, carbon atom undergoes sp 3 hybridisation. This makes hcn a linear molecule with a 180° bond angle around the central carbon atom. The four electrons enter into the new four identical hybrid orbitals known as sp 3 hybrid orbitals one each as per hund. N has one sigma bond to c, and the other sp hybrid orbital exists for the lone electron pair. Carbon (atomic number z=6) … Similarly one may ask, how many carbon atoms are sp3 hybridized? The two singly occupied p orbitals can be utilized for bonding to give methylene ch 2, an unstable free. 03.12.2020 · what is an sp 3 hybridized carbon atom.. 03.12.2020 · what is an sp 3 hybridized carbon atom.

This makes hcn a linear molecule with a 180° bond angle around the central carbon atom. Which carbon has sp3 hybridization? Similarly one may ask, how many carbon atoms are sp3 hybridized? This makes hcn a linear molecule with a 180° bond angle around the central carbon atom. Carbon (atomic number z=6) …
This leaves 4 valence electrons which will each overlap with the s orbital of a hydrogen to form a σ (sigma) bond. Both c and n have 2 p orbitals each, set aside for the triple bond (2 pi bonds on top of the sigma). The carbon atom, that forms four sigma bonds, is sp3 s p 3 hybridized carbon. This makes hcn a linear molecule with a 180° bond angle around the central carbon atom. Similarly one may ask, how many carbon atoms are sp3 hybridized? The two singly occupied p orbitals can be utilized for bonding to give methylene ch 2, an unstable free. This leaves 4 valence electrons which will each overlap with the s orbital of a hydrogen to form a σ (sigma) bond. Carbon has 1 sigma bond each to h and n. Which carbon has sp3 hybridization? The four electrons enter into the new four identical hybrid orbitals known as sp 3 hybrid orbitals one each as per hund.. Carbon has 1 sigma bond each to h and n.

This makes hcn a linear molecule with a 180° bond angle around the central carbon atom. Methanethe methane molecule has four equal bonds. 21.09.2021 · both of these atoms are sp hybridized. The two singly occupied p orbitals can be utilized for bonding to give methylene ch 2, an unstable free. The carbon atom, that forms four sigma bonds, is sp3 s p 3 hybridized carbon. 03.12.2020 · what is an sp 3 hybridized carbon atom. This makes hcn a linear molecule with a 180° bond angle around the central carbon atom. Methanethe methane molecule has four equal bonds.

The carbon atom, that forms four sigma bonds, is sp3 s p 3 hybridized carbon... The ground state configuration of carbon is 1s 2 2s 2 2px 1 2py 1. Methanethe methane molecule has four equal bonds. Carbon has 1 sigma bond each to h and n. Similarly one may ask, how many carbon atoms are sp3 hybridized? 03.12.2020 · what is an sp 3 hybridized carbon atom.

03.12.2020 · what is an sp 3 hybridized carbon atom. Both c and n have 2 p orbitals each, set aside for the triple bond (2 pi bonds on top of the sigma). This makes hcn a linear molecule with a 180° bond angle around the central carbon atom. The four electrons enter into the new four identical hybrid orbitals known as sp 3 hybrid orbitals one each as per hund. The six c‒h sigma bonds are formed from overlap of the sp3 hybrid orbitals on c with the 1s atomic orbitals from the hydrogen atoms. This leaves 4 valence electrons which will each overlap with the s orbital of a hydrogen to form a σ (sigma) bond. The two singly occupied p orbitals can be utilized for bonding to give methylene ch 2, an unstable free. The ground state configuration of carbon is 1s 2 2s 2 2px 1 2py 1.. Which carbon has sp3 hybridization?

The carbon atom, that forms four sigma bonds, is sp3 s p 3 hybridized carbon. The six c‒h sigma bonds are formed from overlap of the sp3 hybrid orbitals on c with the 1s atomic orbitals from the hydrogen atoms. This makes hcn a linear molecule with a 180° bond angle around the central carbon atom. Carbon has 1 sigma bond each to h and n. The four electrons enter into the new four identical hybrid orbitals known as sp 3 hybrid orbitals one each as per hund. The two singly occupied p orbitals can be utilized for bonding to give methylene ch 2, an unstable free. 04.04.2019 · the carbon atoms are sp3 hybridized. Carbon (atomic number z=6) … Both c and n have 2 p orbitals each, set aside for the triple bond (2 pi bonds on top of the sigma). The six c‒h sigma bonds are formed from overlap of the sp3 hybrid orbitals on c with the 1s atomic orbitals from the hydrogen atoms.
